'Don't Know On What Basis Vaccines Were Approved, There Is No Data': Dr Pradeep Seth, Virologist
Writer: Rakshitha R
Rakshitha an engineer turned passionate journalist with an inclination for poetry, creative writing, movies, fiction, mountains and seclusion. Not a part of the social process but existential.
India, 5 Jan 2021 1:19 PM GMT
Editor : Shweta Kothari |
A broadcast turned digital journalist, Shweta Kothari heads the newsroom at The Logical Indian. She has previously worked with CNBC and NewsX as a news anchor and senior correspondent. Shweta holds a masters degree in journalism from the university of Sussex, UK and started her career with work placement with BBC in Scotland.
Creatives : Abhishek M
" An engineer by profession, Abhishek is the creative producer of the team, graphic designing is his passion and travelling his get away. In more ways than one, he makes the content visually appealing."
In an exclusive interview To The Logical Indian’s Shweta Kothari, Dr Pradeep Seth, Indian Virologist and former head of microbiology at AIIMS answers questions regarding COVID vaccine approval.
India has approved two home-grown vaccines, Oxford-AstraZeneca's 'Covishield' and Bharat Biotech's 'Covaxin' for emergency use authorisation.
The Drugs Controller General of India (DCGI) granted approval for these vaccines on the basis of recommendations by a COVID-19 Subject Expert Committee (SEC) of the Central Drugs Standard Control Organisation (CDSCO).
The hasty approval has raised questions among the virologists, experts and citizens whether the scientific procedure has been an undercut to approve the COVID-19 vaccines.
In an exclusive interview To The Logical Indian's Shweta Kothari, Dr Pradeep Seth, Indian Virologist and former head of microbiology at AIIMS answers questions regarding COVID vaccine approval.
Q) Both the vaccines have received emergency approvals. The efficacy of Covishield is 70% but we do not know the efficacy of Covaxin. Is that a cause of concern?
Frankly speaking, that is definitely a cause of concern. Nothing about the efficacy of Covaxin has been out in the public domain yet. I have gone through many newspapers but did not find any information on that. The data should have been put in the public domain by now.
Besides, they have tried an old technique which now nobody uses, where the virus genome is inactivated, for raising the immunity. But whatever they have done, it is done in a hurry.
Let me refer to a case in the 1950s where a polio vaccine was administered to children. When the Polio vaccine was tried back then, it was an inactivated vaccine, which was not properly inactivated, after which a lot of children developed polio. Having said this, I only feel scientists could have used a better way to develop the vaccine.
Q) Phase three trials that are underway and there is no data in the public domain. How do you read that? Should that be worrying us or should we be believing what the government has to say?
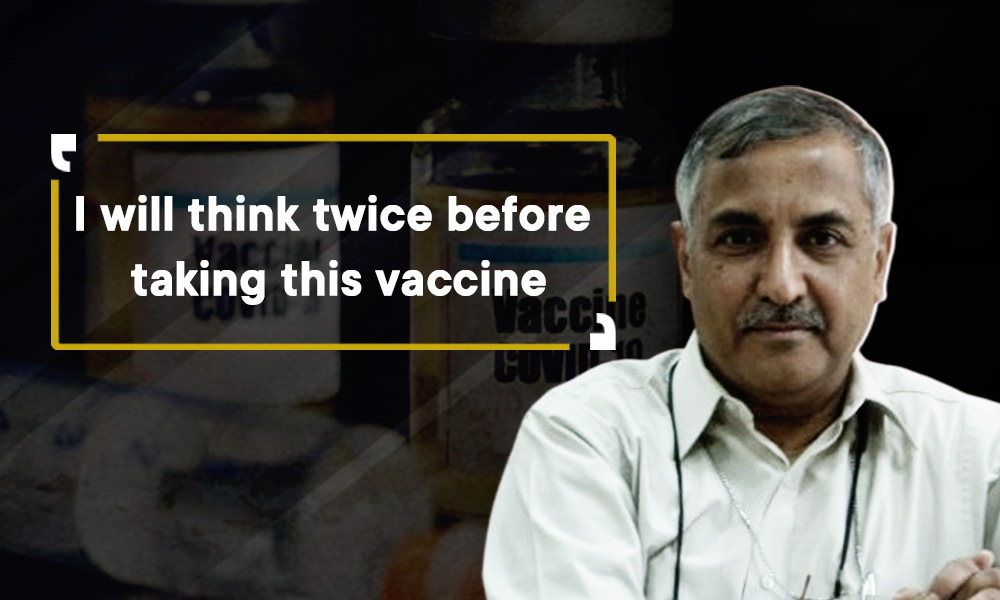
I do not know on what basis the vaccine has been approved, as I have not seen those things/papers yet. But the government committee is a powerful one that comprises of a couple of members whom I know. But on what basis they approved, I do not know. I am a bit confused on this issue. But since there is no data available, I will think twice before taking this vaccine.
Q) As per the information we have received, they are calling it an 'experimental drug' although the emergency approval has now been given. Now, to a lot of people who want to be looking at taking the vaccine, I want to know from you, what does it mean for a drug to be under experiment, and how safe is it for usage?
Whenever such experimental drugs are used for emergency purposes, they are used in circumstances where the complete data is yet to be procured or developed. But given the situation of the pandemic, you have to allow the drugs to be used. For example, Penicillin was allowed in the second world war when it was still under development. It was an experimental drug but it was allowed to be used.
The committee which has approved the COVID-19 vaccines must have seen the data but has not revealed it.
Q) The government has hurried in approving the vaccine, does that mean the science was undercut?
The usual development of the vaccine takes about 5 to 7 years. I have experienced it because I have developed an HIV vaccine which took me almost 7 years. But as it is a case of emergency now, there must have been an undercut.
Q) Then, what could have been done?
The companies should reveal the data instead of confining it to their chest. This is the concern of not just mine but everyone. The companies should do this to gain the trust of people.
Q) We all need the vaccine at the earliest but what we do not need is a vaccine that people do not trust, right? What do you say?
You always develop trust when there is something available in 'writing'. Covishield has brought the data to the public domain. Pfizer and Moderna have also released the data. That type of attitude should be maintained by any vaccine developer.
Q) Many are calling this vaccine a 'nationalism'. We know that our Prime Minister promised that there will be vaccine by August 14, but there was not any. What is your take?
India has got a good population of scientists but why we were not able to produce a vaccine at the earliest? What are the scientists doing? I am 78-year-old now. If I am given money to develop a vaccine, I would still do it. When we can produce good satellites, why not a vaccine? We always thump our chest saying we are the greatest in the world. Why can't we work for a good vaccine?
Q) But many people questioned as to, why they should trust the government or the approval committee. As an expert, what do you say to people?
When we (scientists) ask for funds for any projects, they (government) ask us why and on what basis can the funds be sanctioned? I have been given projects since the start of my career. Until my retirement, I have been given plenty of funds by the government. Even now if I ask for funds, they ask me why I need funds. Don't they trust me?
The government does not sanction funds unless I show my project in publication. Now, this is exactly what we are asking for. The data, where is the data?
Q) What is the way forward? We know emergency approval has been given. What is your expectation from the vaccine developers and the government of India at the moment?
The Serum Institute Of India (SII) has to publish the data. They should not simply rely on England's' study. SII is just reaping the harvest based on England's data. The approval committee should come out and ask the company to release the data on its vaccine's efficacy and safety.
Also Read: Uttar Pradesh: Dalit Girl Kills Self After School Bars Her From Exam
 All section
All section














