A recent social media post shared by Jitendra Chouksey, founder of fitness platform Fittr, has ignited widespread discussion on the cervical cancer vaccine.
Known for his focus on health and wellness, Jitendra highlighted concerns about death statistics and vaccine safety, urging people to question the numbers. His post points out issues with death numbers and vaccine safety, sparking debate amid India’s recent free HPV vaccination drive.
The post quickly drew strong responses from doctors, pathologists, and oncologists, while also resonating with sections of the public. In the process, a complex and important conversation around HPV vaccination, data, and public health communication risked being overshadowed by the intensity of the reactions.
Do We Have Data on Cervical Cancer Deaths?
Jitendra’s post argues that India lacks solid cervical cancer death numbers, unlike road accident stats from police logs. He says claims like “one woman dies every 8 minutes” come from social media hype, not hard facts. This touches a real nerve, as cancer tracking feels less straightforward than traffic reports.
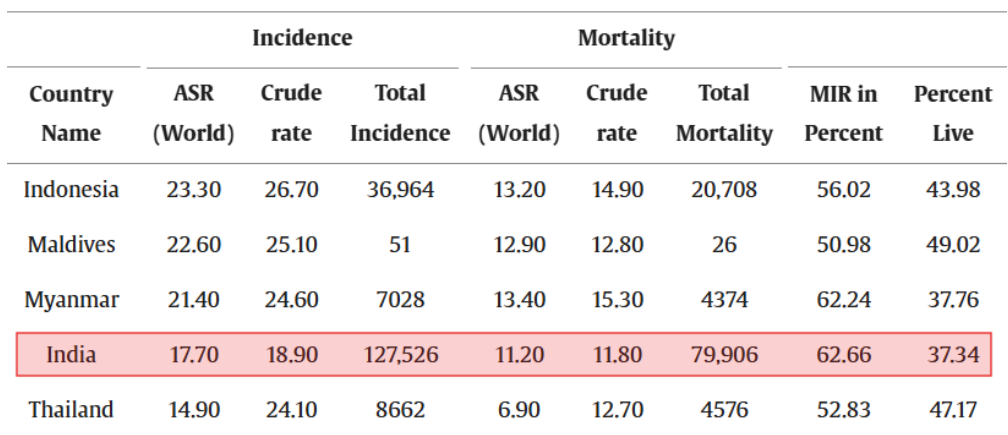
Yet data does exist, mainly as estimates from cancer registries. In India, official data from the Indian Council of Medical Research-National Cancer Registry Programme (ICMR–NCRP) estimates 35,691 cervical cancer deaths in 2023, up from 34,806 in 2022. By contrast, global estimates from GLOBOCAN 2022 place the toll at about 79,906 cervical cancer deaths per year, reflecting a more aggressive attempt to correct for under‑counting in countries with weak death‑certification systems.
Cervical cancer remains one of the most preventable cancers in the world. The HPV vaccine, which targets high-risk strains of the Human Papillomavirus, particularly types 16 and 18 that account for around 70% of cases globally, is supported by decades of clinical research.
These figures are not perfect and not without limitations, but they represent the best available attempts to understand disease burden using existing data sources.
Women are being diagnosed. Lifetimes are being derailed. In many instances, particularly in tier‑2 and tier‑3 cities, women reach hospitals only when the disease is advanced, because screening (like VIA) often misses early lesions and awareness remains patchy.
Have HPV Vaccines Ever Been Tested Against Actual Cancer?
Chouksey’s post correctly notes that no phase‑3 randomized controlled trial (RCT) has used invasive cervical cancer as the primary endpoint. Instead, trials measure CIN2 and CIN3 lesions, cervical intraepithelial neoplasia stages that are early, abnormal changes in cervical cells.
CIN2/CIN3 are widely accepted early warning signs of cancer risk, even though not all lesions progress to cancer. Many CIN2 cases regress on their own; only a subset progress to CIN3; and not all CIN3 lesions become cancer.
But the vaccine’s job is not to treat existing lesions. It is to prevent HPV infection in the first place, typically given to girls aged 9-14, ideally before sexual activity. Large trials like FUTURE II (over 12,000 women) found that Gardasil reduced high‑grade CIN3+ lesions by about 98% among HPV‑naïve women. The PATRICIA trial (over 18,000 women) showed that Cervarix reduced the risk of CIN3+ by about 93%.
A Swedish study, later extended with an 18‑year follow‑up and published in The BMJ, found that “individuals vaccinated before age 17 had a substantially (79%) lower risk of cervical cancer compared with the unvaccinated group, with sustained protection (77% lower risk) for 13-15 years after vaccination.”
Is HPV Vaccine Safe?
Medical researchers are weighing contrasting evidence on HPV vaccine safety. A study called Real-world safety of HPV vaccines over 18 y, analyzed 76,575 reports “finding no association between quadrivalent HPV vaccination and demyelinating diseases of the central nervous system.”
In contrast, Jorgensen’s Benefits and harms of the human papillomavirus (HPV) vaccines identified an exploratory 49% increase in serious nervous system disorders, alleging a “high risk of bias” in original clinical trials. While both studies acknowledge a reduction in cancer precursors, they diverge on rare side effects.
Now, here’s the thing: no vaccine is 100% effective or completely free of side effects. Even commonly used medicines can have mild to severe adverse effects. That, however, does not make them ineffective. Similarly, taking the HPV vaccine does not guarantee that someone will never develop cervical cancer, it significantly reduces the risk.
Ultimately, the decision to get vaccinated is a personal one that should be made after considering both the potential benefits and possible side effects. Consulting credible medical guidance and evaluating the evidence can help individuals make an informed choice for themselves or their children.
It is also important to look at how these findings are derived. Jorgensen’s study, shared by Chouksey, reports this increased risk based on an exploratory analysis with a four-year follow-up. The other study, on the other hand, draws on 18 years of data from the Vaccine Adverse Event Reporting System. So while these findings shouldn’t be ignored, the question remains: can they be treated as absolute, established truth?
India’s Poor Data Infrastructure
Here is where we need to be honest and uncomfortable. Chouksey went to the ICMR website and government data portals, the official, go‑to sources that policymakers and regulators rely on and pulled up cervical cancer death data.
What he found appeared limited, with gaps in publicly accessible, up-to-date data. Patchy numbers from earlier years, showing “numbers growing at a fix percentage every year. They’ve been synthesised,” Jitendra points out.
Now, if one were to reconstruct the prevailing medical‑community mindset, it might sound like this: “Even if the data systems are imperfect, we know the disease is real and the vaccine works. Poor data should not become a pretext to reject sound science.” That reasoning is medically justifiable but institutionally dangerous. Why?
Because ICMR–NCRP and government health portals are not just websites. They are the single most authoritative reference point for health policy in this country. When a regulator, a policymaker, a journalist, or a researcher wants to make a data‑backed argument about cervical cancer, that is where they go. That data is the foundation on which decisions are made, budgets are allocated, and public health campaigns are built.
If that foundation looks dusty, if the dashboards are hard to read, timelines are unclear, and international readers find only archived PDFs instead of intuitive visualisations, it is not a minor inconvenience. It is a systemic failure.
This Is Not a Capability Problem. It’s a Priority Problem.
The government can do this, we’ve seen it before. India’s handling of COVID‑19 data, for all its early chaos, eventually became a case study in mobilisation.
The vaccination dashboard, real‑time tracking, district‑level granularity, all of it was built from scratch, under pressure, with no playbook. The government showed, in the most adverse conditions imaginable, that it can collect, process, and communicate health data at scale.
So the question is not ‘Can we maintain updated, reliable cervical cancer surveillance data?’ The question is: ‘Have we decided that it matters enough?’ The answer, evidently, is not yet.
Why Doubts Linger in India
Public hesitation persists for several reasons:
- Past controversies, such as the 2010 HPV trial in Andhra Pradesh, where seven deaths occurred post‑vaccination, later deemed unrelated to the vaccine by investigators, have damaged trust.
- Additionally, limited awareness about cervical cancer and its prevention, gaps in access to regular screening services, and uneven health outreach across regions also play a role.
As a result, many cases in India are still detected at later stages, highlighting the importance of both awareness and early intervention.
Balancing Prevention and Caution
Chouksey’s post, shared from his wellness perspective at Fittr, brings essential questions to a vital public health moment in India. With the free HPV vaccine program now underway, his call to scrutinize death estimates, trial methods, and benefit-harm math spotlights real gaps, like limited registry coverage and data lags, that no one should ignore. Official figures from ICMR and WHO paint a serious cervical cancer picture, while global trials and data suggest vaccines offer strong upstream protection.
The debate underscores a key truth: health choices deserve full transparency, especially for parents eyeing shots for their daughters. The discussion ultimately highlights the importance of clear communication in public health.
Bridging the Data Gap with Concrete Fixes
Differences between estimates from GLOBOCAN and ICMR–NCRP largely reflect methodological approaches to addressing underreporting. While global models adjust upward to account for gaps in data, national registries tend to rely on reported cases, leading to more conservative estimates.
Bridging this gap requires strengthening India’s health data systems. This could include expanding cancer registries beyond urban centres, improving cause-of-death reporting, and integrating data across registries and hospitals. Greater transparency in how estimates are generated and communicated may also help build public confidence and reduce confusion around differing figures.
Editor’s Note: This article is part of The Logical Take, a commentary section of The Logical Indian. The views expressed are based on research, constitutional values, and the author’s analysis of publicly reported events. They are intended to encourage informed, responsible dialogue around health communication and data transparency, and do not seek to target or malign any individual, institution, or community.
Disclaimer: The decision to receive the HPV vaccine is a personal one and should be made after considering both its benefits and potential risks, ideally in consultation with qualified medical professionals. This article has been revised and updated after publication to refine certain technical details and ensure factual accuracy.













